3D Organoids and the study of lung disease
by Sara Nolbrant, MSc, Lund University
To be able to more specifically study human disease, scientists have been growing human cells in culture dishes for over a century. These cell cultures are classically grown as 2D sheets. This means that the system is very simplified in comparison with the body, where different kinds of cells intermingle and build up complicated architectures. The loss of complexity when growing cells in a dish makes it difficult to draw conclusion on how the cells behave in the body. So what if instead of growing cells in 2D we do it in 3D? In a way that more resembles the cells natural habitat.
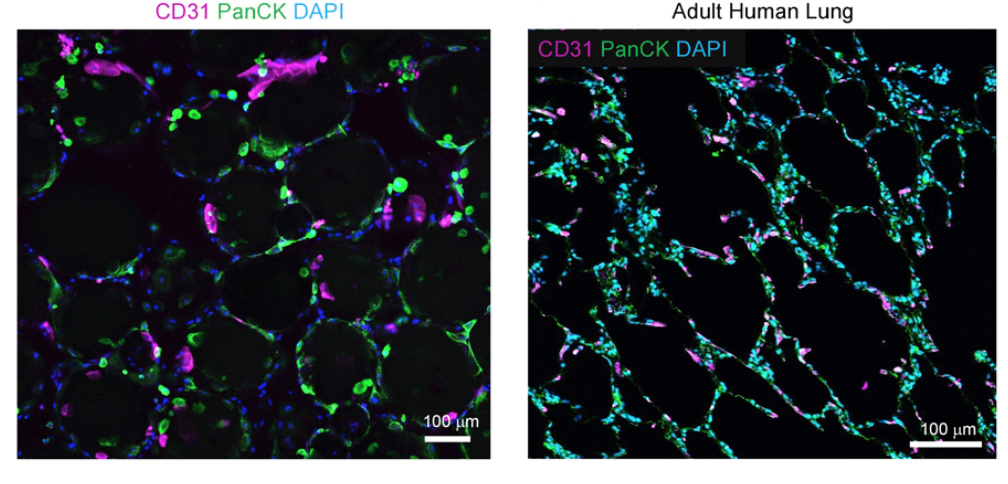
Image credit: UCLA Broad Stem Cell Research Center
Since early 2010’s the techniques of creating lab-grown organ buds, so called ‘organoids’, have become more and more sophisticated. These organoids are miniature organ-like structures that can be maintained in the lab, and scientists are now able to grow organoids resembling many different tissues. From brains and hearts to kidneys and livers (only to mention a few). Successful 3D organoids, or organ mimics, need to meet a few criteria. They must replicate the organ’s anatomy, contain the same types of cells as the organ does and organize these cells into their correct ‘compartments’ - which build up the structure of the organ. These 3D organoids open up a range of new possibilities when it comes to studying human development and disease and for the development of personalized medicine. The ability to create lab grown organoids will be particularly valuable for conditions which are hard to model, with our existing animal models or classical 2D cell cultures.
A good example of this is lung disease. Lung diseases, such as chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF), represent a major cause of illness and death worldwide. When the population age, as is the case in today’s society, even more people will be affected. Given the fact that many lung diseases are currently without any cure, there is a desperate need for new effective therapies.
In a study recently published in Stem Cells Translational Medicine, researchers at the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA set out to develop a new system for building lung 3D organoids to model lung disease (see image). To create these organoids, the UCLA team used tiny gel beads that were coated in a way that made cells stick to them. These beads resembled the lung alveoli, the small lung-bubbles where the air can move between the lungs and the blood. When the cells stuck to the beads, these could then aggregate and self-organized into organoids which looked very similar to lung tissue and contained many different cell types.
To prove that the system could be used for personalized medicine, the researchers also used so called induced pluripotent stem cells (or iPSCs). This type of stem cell can be generated by taking, for example, a skin cell and revert it back to a stem cells state, where it has the potential to become any type of cell in the body (if given the right signals). These stem cells were then instructed to mature into lung cells and could be incorporated into the organoids, thereby making the organoids individualized, or patient-specific. These patient-specific organoids could be useful in predicting how a specific person's cells will respond to a particular drug. Or to investigate the biological cause of disease.
Next, the team wanted to test if their lung organoids could be used to model disease. Idiopathic pulmonary fibrosis (IPF) is a fatal disease where the lung tissue becomes scarred and stiff. To model IPF, the researchers treated the organoids with a molecule called ‘TGF-β1’, which is known to play a role in the scarring of tissue. When this molecule was added, the organoid changed in appearance and started to develop scar-like structures, similar to those seen in people affected by the condition. This is the first time anyone has been able to model the pathological hallmarks of IPF in a dish, since these changes do not appear in regular flat 2D cultures. The possibilities of finally being able to model IPF in the lab open up many doors for future studies.
Lung diseases, such as IPF, where there is a lot of variation between people will have a lot to gain from an individualized model to test which drug will work best for that particular person. Scientists could take the patient’s own cells and use them to to create personal organoids. These could then be used to test different potential drugs, in order to generate customized treatments. More research will be required before these progresses can be translated into something concrete for patients. However, this proof of principle research demonstrates the promising outlook for this avenue of stem cell research.
Last updated: