Chronic liver disease: how could regenerative medicine help?
Chronic liver disease is the fifth biggest killer in the EU. Once serious damage has been done to the liver, it loses the ability to repair itself and this is a life-threatening problem. The only treatment currently available is a liver transplant. Could regenerative medicine help?
The liver is the only internal organ in the human body capable of regenerating itself after being damaged.
In chronic liver disease, damage to the liver over long periods of time leads to the accumulation of scar tissue that limits the ability of the liver to function and repair itself. This disease is the fifth largest killer in the EU and presently can only be treated with liver transplants.
Researchers have successfully used embryonic stem cells and induced pluripotent stem cells to make new liver cells in laboratories, which may potentially be used to treat liver disease in the future.
Researchers want to learn how stem cells in the liver are able to regenerate liver tissue. It may be possible to develop treatments that harness the natural ability of liver stem cells to regenerate the liver. Treatments using pluripotent stem cells to create new liver cells for transplantation into the liver are being developed.
Researchers are also developing bio-artificial liver devices and methods to bioengineer livers for transplantation.
Studies are also currently exploring if a person’s blood cells might be used to generate a specific type of macrophage cells that will remove scar tissue from the damaged liver.
There are not enough suitable organ donors to match the demand of liver transplants for liver cirrhosis. Also, liver transplants require patients to take immunosuppressants to prevent transplant rejection.
Many stem cell treatments could potentially avoid the issue of immune rejection, however additional work must still be carried out to make sure that stem cell treatments, particularly pluripotent stem cell treatments, create mature and functional liver cells that are safe for transplantation in large enough quantities.
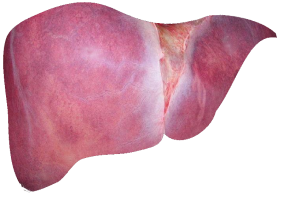 The liver is the largest internal organ of the human body. It does many jobs, including removing toxins from the blood, helping to digest food, and fighting infections. It is the only organ in the body that can regenerate itself after damage.
The liver is the largest internal organ of the human body. It does many jobs, including removing toxins from the blood, helping to digest food, and fighting infections. It is the only organ in the body that can regenerate itself after damage.
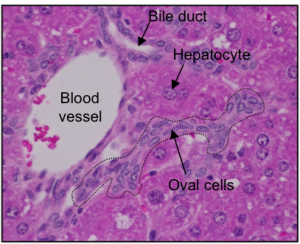
The cells that do the work in the liver are called hepatocytes. On average, each hepatocyte lives for around 200 to 300 days. In a healthy liver, hepatocytes can divide to make copies of themselves. This means they can replace the cells that die and can even repair some kinds of damage. If the liver is severely injured, another type of liver cell may come to the rescue: these cells are called hepatic progenitor cells (HPCs). HPCs are thought to be the liver’s resident stem cells and have the potential to make new hepatocytes. However, scientists are still investigating exactly what HPCs are, how they work and how we can make them produce hepatocytes more efficiently.
In chronic liver disease, a lot of liver damage happens over a long period of time. The normal repair processes are impaired and scars are formed in the liver (also called cirrhosis). The only currently available treatment for patients with cirrhosis is a liver transplantation. However, transplants are expensive, the process requires lifelong immunosuppression and there are not enough organ donors to treat all patients. Alternative therapies must therefore be found for patients with liver cirrhosis.
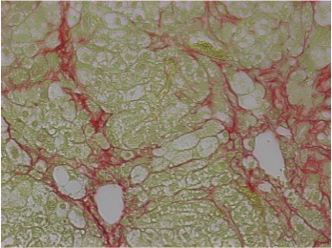
Other than a liver transplant, there are currently no conventional treatments or stem cell treatments for chronic liver disease that are approved for clinical use. However, in the long term, stem cells might provide the ability to develop new approaches for treatments:
- Preliminary clinical trials have seen some success in transplanting new hepatocytes into a patient’s liver. However, there are several large obstacles to this approach. Attachment (or engraftment) of these new cells into the liver is often very low. Also, transplanted cells are susceptible to attack / rejection by the immune system (transplant rejection). The biggest issue for this treatment is that this procedure requires large numbers of new cells for transplant, which are not readily availabile. On this last issue, stem cells, such as induced pluripotent stem cells (iPSCs), combined with new culturing techniques could offer significant help in creating enough new hepatocytes for transplants. Using iPSCs to create new hepatocytes may also help avoid attack by the immune system, since iPSCs can be made from cells taken from a patient, such as their skin cells. However, there are still a number of fundamental questions that must be answered before hepatocyte transplants using stem cells can be used clinically. For example, it will be important to find out if stem cell-derived hepatocytes are safe to treat patients and are as functional as normal hepatocytes.
- Researchers are working to identify liver stem cells more precisely and understand how they might be used to treat patients. Researchers are currently working towards establishing new techniques to identify, isolate, and expand liver stem cells from donor tissue for transplantation.
- Bio-artificial livers are an area of growing research that combine technology with biology to create a device similar to a dialysis machine that contains living hepatocytes. In theory, the cells in this device could remove toxins, produce essential proteins for the body (such as albumin for blood serum), and provide other vital roles of a healthy liver. As with hepatocyte transplant treatments, obtaining large enough numbers of human hepatocytes to live in these bio-artificial livers is a major obstacle. Researchers are attempting to address this issue by using hepatocytes from animal donors (such as pigs) or human stem cells (such as iPSCs) to grow large numbers of fully functional hepatocytes. Clinical results in studies examining bio-artificial livers have been mixed, with some showing no benefits over current treatment methods. However, next generation technologies for growing cells in these bio-artificial livers may greatly advance the quality of the cells, cell metabolic activity and overall effectiveness of these devices for treatment.
- Research is also advancing technologies to bioengineer whole organs, including the liver. Bioengineering a liver is complex, requiring the ability to assemble extremely large numbers of functional hepatocytes into specific three-dimensional structures with other types of cells, such as cells that create the vascular system. Researchers have determined how to create scaffolds for hepatocytes to grow on and direct their general arrangement, but there are still large challenges. Once again, having large numbers of human hepatocytes is a problem. In addition, it is still not clear how to incorporate a vascular system and other types of cells into these bioengineered livers. Stem cells, particularly iPSCs, offer the potential for creating the large numbers of hepatocytes needed, but researchers have to make sure that these cells have the appropriate levels of metabolic activity and will not continue to multiply once in the body. Unregulated growth in stem cell-made hepatocytes has potential to lead to tumours. Bioengineering the liver with stem cells is an ambitious goal, but has the potential to avoid or solve many problems associated with other treatment methods. Such a technology would completely replace the damaged liver with a new liver and potentially avoid attack by the immune system if made with iPSCs.
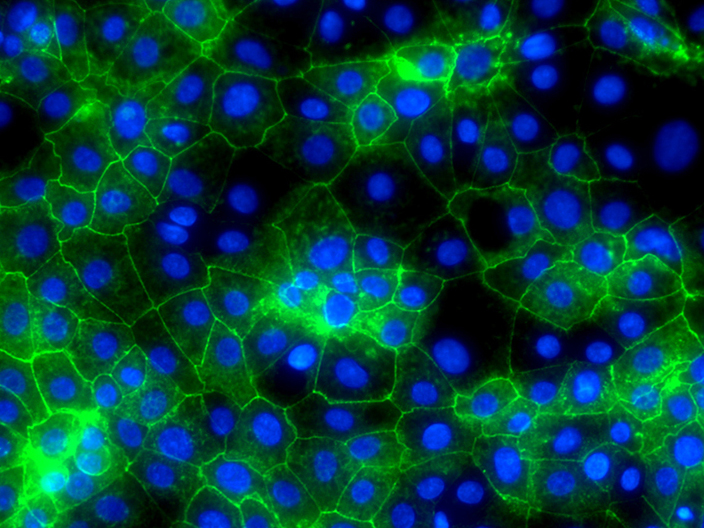
Another route to new treatments might be to use cells made from a patient’s own blood to help repair damaged liver tissue. Cells in the blood called ‘blood monocytes’ can be grown in laboratories to produce large quantities of another type of cell in the blood called macrophages. When tested in mice with damaged livers, macrophages show a beneficial role in regenerating liver tissue and removing scar tissue in the liver. When scarring is reduced, the liver is able to work better. Recent work has shown that macrophages can secrete enzymes that break down scar tissue directly, and also help remove cells that produce the scar tissue in the first place. Human macrophages are now being tested in patients with cirrhosis in clinical trials to test how safe and effective they are. Macrophages are an attractive cell therapy because they can be derived from a patient’s own blood cells and therefore would not be rejected by the immune system, as sometimes happens with organ transplants.
The British Liver Trust - information for patients
Liver Good Life - an animated film about the liver
The European Association for The Study of the Liver - information on EU policy
Centre for Regenerative Medicine - lab studying liver stem cells and regeneration
Centre for Regenerative Medicine - macrophage cell therapy for liver cirrhosis
This factsheet was first created by Caroline Pope.
Reviewed in 2010 by David Hay, David Tosh and Clare Blackburn.
Reviewed and updated in 2015 by David Hay.
Edited in 2018 by Ryan Lewis.
Reviewed and updated in 2019 by Philip Starkey Lewis and Benjamin Dwyer.
Cell images by Caroline Pope, David Hay and Luke Boulter.