Neural stem cells reveal how Zika virus can cause microcephaly and a potential drug treatment
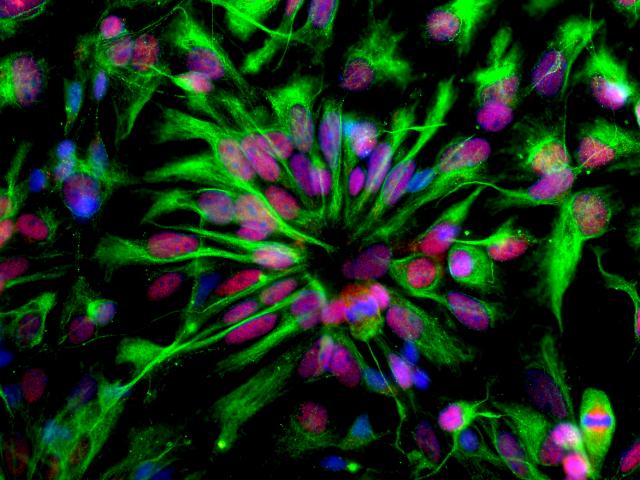
Image: Marco Onorati, Neuroscience Dept, Yale School of Medicine.
By Marco Onorati
A team of Yale researchers has discovered that infection by the Zika virus (ZIKV) stops neural stem cells dividing in the developing human brain, thereby causing the birth defect microcephaly. The ZIKV does this by diverting a key protein necessary for neural stem cell division. The researchers also reported that antiviral nucleoside analogs, including the FDA-approved drug Sofosbuvir (normally used to treat Hepatitis C virus infection), inhibit ZIKV replication and protect human neural stem cell from death.
What is the idea behind this study?
ZIKV, a flavivirus closely related to dengue and yellow fever viruses, was discovered in the Zika forest of Uganda in 1947 and reported sporadically in Africa and Asia. ZIKV has expanded its geographic range dramatically to the Americas and the Pacific areas since 2007. More recently, South America has experienced a ZIKV outbreak, with around 30.000 cases of ZIKV infection in Brazil. While ZIKV infection was believed to cause a mild illness, associations between neurological disorders, including Guillain–Barré syndrome (a disease causing muscle weakness and paralysis) has been proposed recently. More prominently, ZIKV infection during pregnancy is associated with, and likely causative for, severe fetal abnormalities including microcephaly, a neurological condition in which the infant's head is abnormally smaller.
The human brain is a complex organ that, as a result of its extended development, is susceptible to a host of genetic and environmental insults that can severely affect its formation and eventually function. Primary microcephaly results mainly from the depletion of neural stem cells, the founder or progenitors of all the cells present in the mature brain, i.e. neurons and another important cell type in the brain, glia.
What did the study show?
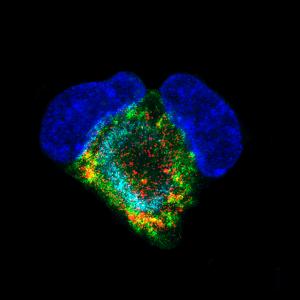
Image: Marco Onorati, Neuroscience Dept, Yale School of Medicine
In this work, the authors describe a new type of neural stem cell, called neuroepithelial stem (NES) cells. NES cells are derived from the earliest population of neural stem cells present during the first stages of brain development. These cell populations serve as the stem or progenitor cells for neurons and glia. They also provide scaffolding that help guide emerging neurons to the correct place in the brain. The researchers used these NES cells (see image above) to recreate the brain disorders associated with ZIKV infection in the lab. This is termed disease modeling.
By comparing NES cells and postmortem human brain tissue in the context of ZIKV infection, they found that ZIKV preferentially infects the neural stem cells and, to a lesser extent, mature neurons. In particular, ZIKV infection in human neural stem cells results in them being unable to undergo mitosis, the process by which a cell divides and proliferates. The impairment ends up with cell death. A detailed analysis shows that ZIKV diverts a protein (called pTBK1) from its primary job of organizing cell division. Instead, pTBK1 is redirected toward mitochondria, the cell’s power pack, where it helps initiate an immune response. Lacking the pTBK1 protein at the site of cell division, cells die instead of forming new brain cells, resulting in microcephaly (Image 2 on the right shows a cell infected with ZIKV). By analyzing postmortem ZIKV-infected human brain tissue, the authors also found that the infection caused a scaffold disorganization, thus impairing the correct migration of the neurons in the brain and contributing more to the microcephalic phenotype (Image 3 below shows disorganised brain tissue, ZIKV is shown in green).
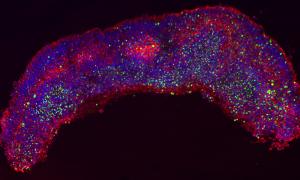
Image: Marco Onorati, Neuroscience Dept, Yale School of Medicine
Further work by the authors suggested that pTBK1 may also contribute to microcephaly associated with other common congenital infections causing microcephaly (such as cytomegalovirus or CMV).
What does this mean for people affected by ZIKV infection?
After identifying the mechanism linking ZIKV and microcephaly, the researchers aimed at a second goal: providing a potential therapeutic avenue. There is an urgent need to identify therapeutic approaches for halting ZIKV infection, especially in pregnant women. The authors screened some drugs, known to be potent antivirals. They identified two nucleoside analogs, including the FDA-approved drug Sofosbuvir, that inhibit ZIKV infection and therefore stop the death of the NES cells.
As a concluding remark, Sofusbuvir is not, at present, a medication that people currently facing Zika should use. While Sofusbuvir is approved for the treatment of chronic hepatitis C virus infection, it is too early to recommend the use of Sofosbuvir in pregnant women. Even if animal studies found no effect of Sofosbuvir on fetal development, there have been no adequate studies of Sofosbuvir in pregnant women and it is currently used with great caution in treating pregnant women with chronic hepatitis C infection. This study would needs to be repeated in clinical studies before we could be sure Sofusbuvir might be a safe and effective ZIKV treatment.
Further information and links
- This summary is based on the original article 'Zika Virus Disrupts Phospho-TBK1 Localization and Mitosis in Human Neuroepithelial Stem Cells and Radial Glia' by Marco Onorati et al., Cell Reports, September 2016. This article is open access.
- EuroStemCell article on 3D mini-brains and how they have been used to explore how the ZIka virus infects the brain.
- EuroStemCell article on neural stem cells and mini-brains also called organoids.
Acknowledgements
Written by Marco Onorati, reviewed by André Sousa and edited by Jan Barfoot. All images are used by kind permission from Marco Onorati, Neuroscience Dept, Yale School of Medicine.
Last updated: