In the Spotlight: The First Gene-Edited Babies
The world's first gene-edited babies were born in 2018 and caused a global controversy. Kirsty Ferguson examines why.
The news
At the end of the decade it was announced that He Jiankui was to be fined 3 million yuan (€390,000) and sentenced to three years in prison for disrupting medical order. He, previously an associate professor at the Southern University of Science and Technology in Shenzhen, China, announced the creation of the first ‘gene-edited’ babies in November 2018. Twins, Lula and Nana, were born following gene-editing of embryos created by in vitro fertilisation (IVF), with a third gene-edited baby also confirmed at the start of 2020.
How was the gene-editing performed?
Seven heterosexual couples were recruited by He’s team for IVF, in which the fertilised embryos were gene-edited prior to implantation into the mother.
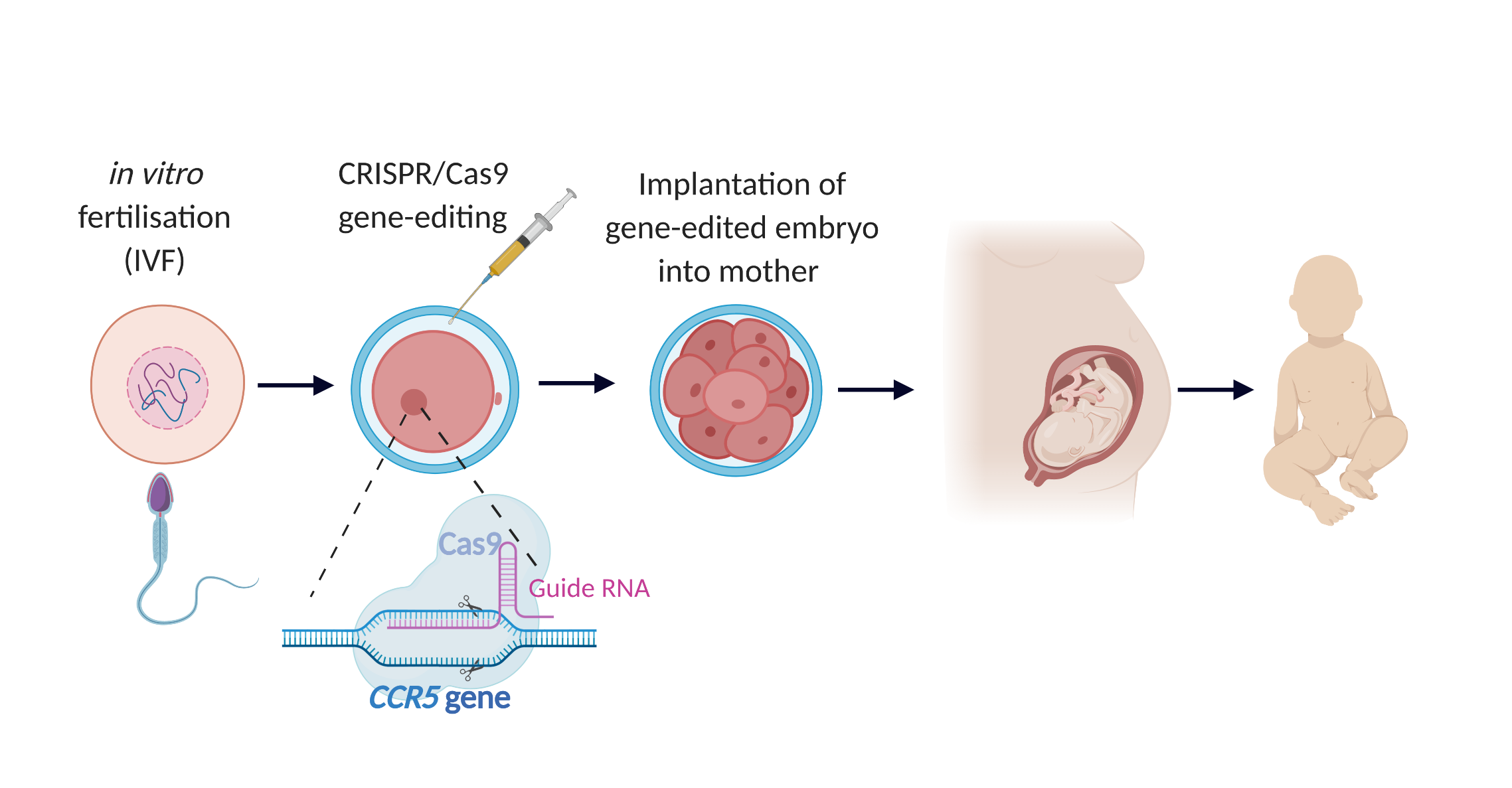
All the recruited men were HIV-positive whereas the women were not; while safe and effective methods exist to prevent the spread of HIV from the father to the embryo, such as sperm washing, the reported goal of the gene-editing was to render the child immune to HIV infection during their lifetime and free from any associated stigma. A gene called CCR5 encodes a molecule that allows HIV to infect immune cells. This gene is naturally mutated in some people, conferring HIV resistance – without CCR5 an individual cannot be infected by HIV. CCR5 was therefore chosen to be disrupted by He’s team using a technology called CRISPR/Cas9 (pronounced ‘crisper’). First used in the lab seven years ago, CRISPR/Cas9 acts like molecular scissors, enabling scientists to precisely modify DNA using a specially-designed molecule to guide a DNA-cutting enzyme (Cas9) to a gene of interest.
Why did this spark a global outcry?
CRISPR has revolutionised research in recent years. The ability to precisely modify DNA is enabling scientists to build models of human diseases in a dish and investigate the potential to alleviate genetic diseases. However, this technology comes with numerous ethical considerations. While it is lawful in some countries to gene-edit embryos for research under strict regulation, many countries, including all EU member states and China, prohibit the genetic modification of human embryos or germ cells (sperm or eggs) for use in assisted reproduction. He’s work disobeyed this and, as said by the court in Shenzhen, “crossed the bottom line of ethics in scientific research and medical ethics”.
One of the main promises of CRISPR is in gene-editing therapies; these involve taking a person’s ‘somatic’ non-reproductive cells that carry a disease-causing mutation, editing the DNA to repair this mutation, and returning healthy cells into the body to eradicate the disease. Such therapies could help to treat genetic diseases such as cystic fibrosis and sickle cell anaemia. However, unlike somatic cells, a gene-edited embryo goes on to make all cells of body, including germ cells. This means the edited DNA has the potential to be passed on to future generations from Lula and Nana. To add to this problem, He’s gene-editing strategy did not create the exact disruption to the CCR5 gene known to confer HIV resistance (termed delta32); rather, other mutations were generated, of which the effects are unknown and unpredictable. CRISPR can also have unwanted ‘off-target’ effects, when DNA is cut in the wrong place; the children could therefore have potentially acquired additional DNA edits, leading to unknown health consequences. As effective methods exist to minimise the risk of HIV exposure, the research of He’s team not only broke the law but also unjustifiably risked the children’s health.
The future of gene-editing
In March 2019, an international group of eminent scientists, including the pioneers of CRISPR technology, called for a five-year global moratorium on the clinical use of heritable DNA editing to make genetically-modified humans. This, they say, would enable the establishment of an international framework and necessary discussions regarding technical, societal, ethical issues. Other scientists worry that a blanket moratorium may hamper credible efforts to advance a field that ultimately has the potential to spare humankind of diseases with known genetic cause. All agree, however, that heritable DNA editing is currently an unjustifiable intervention. As highlighted by He’s work, it requires strict regulation according to a global altruistic consensus agreed by scientists, ethicists, philosophers, policy-makers, and the public. Gene-editing can ultimately affect the entire human species and must therefore occur with transparency and respect for all.
Find out more
This case
China jails ‘gene-edited babies’ scientist for three years
He Jiankui defends ‘world’s first gene-edited babies’
Genome-edited baby claim provokes international outcry
CRISPR and its potential therapeutic uses
CRISPR – changing the gene editing landscape
Quest to use CRISPR against disease gains ground
Allele specific repair of splicing mutations in cystic fibrosis through AsCas12a genome editing
Ethical considerations
The ethics of changing genes in human embryos
Pro and con: Should gene editing be performed on human embryos?
Policy and regulation
Genome editing: scientific opportunities, public interests and policy options in the European Union
The response of the scientific community
Adopt a moratorium on heritable genome editing
A case against a moratorium on germline gene editing
New call to ban gene-edited babies divides biologists
Acknowledgements
Written by Kirsty Ferguson. Reviewed by Catarina Martins Costa and Jan Barfoot.
This is part of a series in which researchers reflect on regenerative-medicine-related stories in the media. This series was initiated by a group of science writers who attended the Hydra European Summer School on Stem Cell Biology and Regenerative Medicine