Do we still need research on human embryonic stem cells?
 By: Austin Smith and Clare Blackburn
By: Austin Smith and Clare Blackburn
Can induced pluripotent stem (iPS) cells replace human embryonic stem (hES) cells? One day this may be the case, but for the foreseeable future, side-by-side research on both types of stem cell is needed.
Facts at-a-glance
We know:
- Pluripotent stem cells are not all the same - mouse ES cells and human ES cells are not equivalent
- Mouse ES cells are more consistent, easier to grow, and easier to differentiate
- Clinical grade human embryonic stem cell lines already exist
- How to make a few specialised cell types from human pluripotent stem cells
We think:
- iPS cells will be an important tool for drug development
- Use of iPS cells in the clinic will require much more research and understanding
- Human pluripotent stem cells equivalent to mouse ES cells may be isolated in the next few years
We do not yet know:
- How to establish the identity and safety of human iPS cells
- How to direct specialization of pluripotent stem cells into many different adult cell types
- How to ensure the cells do not undergo unpredictable genetic changes when grown long-term
There are three key reasons for continuing to study human ES cells:
1. Safety and function
ES cells are essentially normal human embryo cells, whereas iPS cells are created by reprogramming adult cells using molecular engineering. These laboratory manipulations have not yet been perfected and can produce abnormal, potentially unsafe cells. iPS cells are definitely promising tools to investigate disease mechanisms and develop new drugs in the laboratory, but many more careful studies are needed to determine whether they will be safe to inject into people. New technologies are also required to produce fully functional adult cells from iPS cells. Human ES cells therefore provide an essential standard for iPS cell safety and differentiation.
2. Ready for the clinic
Cells that will be transplanted into people must be prepared in a strictly controlled environment to ensure they are not contaminated and are of high quality and purity. Several clinical grade hES cell lines have been developed and are already approved for use in clinical trials. Reaching this point on the road to the clinic is the result of years of research and development. Currently, no iPS cells are approved for clinical use because of the gene manipulation procedures required for their production. It will take time to overcome this hurdle and develop clinic-ready iPS cell lines.
3. The missing human pluripotent stem cell
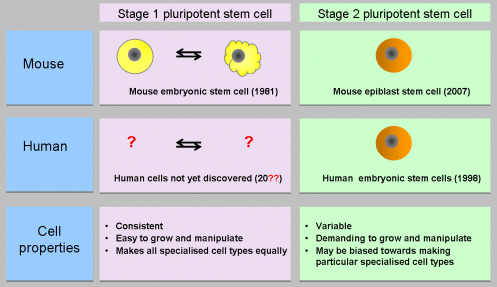 Currently, different human ES and iPS cell lines show a great deal of variability and inconsistency. Mouse ES cells however can be grown under very standardised conditions, with minimum variability.
Currently, different human ES and iPS cell lines show a great deal of variability and inconsistency. Mouse ES cells however can be grown under very standardised conditions, with minimum variability.
Mouse and human ES cells are not equivalent; human ES cells appear to represent cells at a more advanced stage of embryonic development, when they have already begun to specialise. Scientists think that it may be possible to isolate a new type of human pluripotent stem cell that is equivalent to mouse ES cells. These new cells – shown above as ‘stage 1 pluripotent stem cells’ and often referred to as ‘ground state stem cells’ – are predicted to have very consistent properties that will allow reliable differentiation into all types of specialised adult cell. Researchers worldwide are currently investigating how to isolate and grow these human ground state cells.
This has three important implications:
- Finding out how to obtain and grow human ground state pluripotent stem cell requires experiments to modify current human ES cells, and also derivation of new human pluripotent/ES cells using alternative techniques recently developed for mouse ES cells. Only through these experiments can the best possible methods for obtaining human pluripotent stem cells be determined.
- Once obtained, human ground state stem cell lines are likely to replace existing human ES cell lines in medical research and clinical applications.
- Ground state human ES cells will provide an accurate reference for iPS cells which will allow them to be standardised, overcoming one of the major barriers to their use by industry and to regulatory approval for their use in clinical applications.
Further information
References
References to scientific research publications are listed in the downloadable pdf below. Journal subscriptions may be required to access the listed references.
Last updated: