Regenerative Medicine in the UK
by Andrew Webster and John Gardner
The REGenableMED project has undertaken a detailed analysis of the regenerative medicine field and the interplay between business models, measures of clinical utility, patterns of regulatory oversight and clinical workflows within healthcare settings. The results of the research have informed strategies aimed at facilitating the responsible development of effective and useful regenerative medicine products and services.
The project has developed the notion of ‘institutional readiness’ as an analytical framework to help understand how an emerging field of medicine, here regenerative medicine, is more, or less, likely to be adopted in the healthcare system. The progress of innovation projects is often understood in terms of ‘technology readiness’: the extent to which the innovative technology has matured, its risks known, and is 'ready' to be put to use. Yet successful innovation also depends upon institutional readiness, or the capacity and willingness of key pre-existing organisations and networks to adopt, respond to and use novel technologies. Institutional readiness is particularly significant in the developing field of regenerative medicine (RM). For RM to live up to its promise of revolutionising patient care, emerging products must somehow be accommodated within pre-existing regulatory and healthcare organisations that are accustomed to dealing with more conventional medicinal products and services as well as new ‘innovation spaces’ opened up for them. In short, it is very important to distinguish between a technically ‘working’ technology and a socio-technically ‘workable’ technology, that is, one that makes sense and is useable in actual contexts.
A simple analogy here can help: developing an innovation without thinking about the actual context in which it can be used is like trying to put a seven-letter ‘perfect score’ word down on a Scrabble Board when there aren’t in fact enough spaces to do so. Instead, the player has to break-up the word to fit, to make it ‘workable’ in those constraints. It is of course possible in the real world to create some additional ‘innovation spaces’ but these eventually need too to be incorporated into the wide healthcare context and be adjusted accordingly.
Things are made even more complicated because the regenerative medicine field is in fact quite diverse, and moving more, or less quickly on different fronts. The project has identified five different pathways that companies and clinical researchers are developing in taking therapies to the clinic, as shown below.
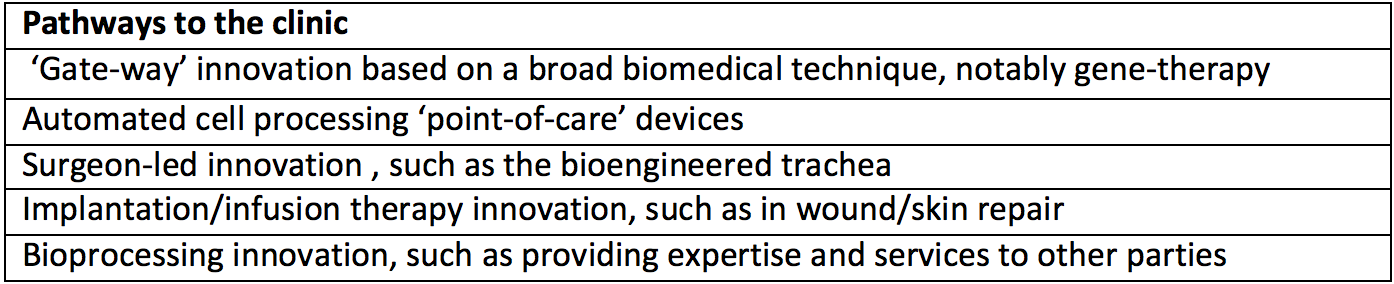
Whatever route is being pursued, however, there are a number of common challenges that will need to be addressed, ones that are key to ‘translating’ regenerative medicine into the clinic. These are outlined below.
There is therefore a need to differentiate activities and so potential for development within the regenerative medicine field. Many applications are still small-scale and at the early trial stage. Most recent investment has been made in the ‘gate-way’ area: there is a strong interest among companies in this area where the potential for treating cancer is seen to be very strong. At present, adoption has been most rapid involving clinician-led surgical procedures whose development may require only minor adjustments to existing clinical practices, such as in limbal stem cell transplantation to restore corneal function, and wound healing and soft-tissue regeneration for leg ulcers typical of diabetics. A key issue is how the administration of cell therapies aligns with existing clinical treatment pathways in specific therapeutic areas.Each of these uses different biomedical techniques, is at different stages on development (some yet to be available), and has to meet different forms of regulation and licensing. They will also be judged differently against existing clinical procedures, and carry very different organisational and economic costs.

Many of these key issues are examined in more detail in the other Themes included in this project summary. Similar challenges can be seen in other areas of emerging biomedical technologies, but underpinning the specific difficulties faced in the regenerative medicine field are three issues. These are:
- the production, stabilisation and use of live cells and tissues,
- their complex and often uncertain regulatory definition,
- their manufacturing/scale-up.
Patient charities
An example of active key stakeholders within this ‘translating’ process are disease focused medical research charities. A growing number of charities are currently invested in the development of RM research at various stages of research from early to phase II clinical trials. These charities can be seen to be in an intermediary position, negotiating a pathway between patients, academia, pharmaceutical industry and investors. From this position, they have the potential to influence patient expectations of RM, whilst being aware of pharmaceutical industry attitudes, and subsequently shaping their own research agendas and priorities. In this way we found that research charities have a part to play in how they both manage hype expectations and set what they regard as being ‘realistic’ future expectations which are crucial to attracting the commitment of their own stakeholders and maintaining an important reputational standing as trustworthy charitable foundations.
To conclude, the adoption of RM therapies will require major, long-term infrastructural commitment to new ways of working within the healthcare system, new skills and new ways of aligning clinical demand and the supply of diverse therapeutic products. More generally, the emergence of complex advanced therapies and combination and borderline products (including medical devices and diagnostics), which do not have a clear regulatory and commercial route to clinic, highlights the need for progressively adaptive regulation that can evolve with emerging scientific knowledge and new technologies.