Regenerating The Thymus
Our bodies need an immune system to fight off infections throughout our lives. The thymus plays an important role in the immune system, but gets smaller and less effective with normal aging and can also be damaged by some medical treatments. How could stem cell research help us replace a lost or damaged thymus, and when would this be useful?
What do we know? ▼
Our immune system fights infection with a specialised type of white blood cell called the ‘T cell’, which are made in an organ called the thymus. The thymus is therefore essential for a healthy immune system.
As we age, the thymus degenerates - becoming smaller and less able to produce the T cells necessary to fight infection.
Currently, there are very few therapies available for improving or restoring thymus function.
However if scientists were able to regenerate the thymus, this could help to boost the immune system and lead to treatments for a number of conditions affecting the thymus, such as DiGeorge Syndrome.
What are researchers investigating? ▼
Given how essential the thymus is for a healthy immune system, scientists are currently trying to understand how they can use stem cells to repair and regenerate this organ.
Scientists have shown that when you take thymus ‘progenitor cells’ (stem cell-like cells), from a mouse and transplant into a mouse that lacks a thymus, the cells go on to make a fully functioning thymus.
Currently, researchers are investigating:
- How to engineer thymus tissue in the lab for transplantation and
- Ways to encourage the thymus to regenerate itself after it has been damaged or has degenerated with age.
What are the challenges? ▼
Much of the current research investigating stem cells as a potential tool to regenerate and repair the thymus has been performed in mice. Discovering whether these findings also work in humans is the current and future big challenge for scientists. For example, before scientists can investigate whether human thymus progenitor cells can create a functional thymus when transplanted, they first need to work out how to grow thymus progenitor cells in the lab and make enough of these cells for transplantation.
About the thymus
The thymus is the organ in which a type of white blood cell, called T cells, are made. Without T cells, we aren’t protected from infections, and can’t control abnormal cells - such as cancer cells - efficiently.
Normal aging causes the thymus to shrink, and it can also be damaged by genetic conditions, chronic infections, and some medical procedures.
If the thymus does not work properly, we can end up with too few T cells to protect ourselves – this is called immunodeficiency – or we can produce T cells that attack our own body – which is called autoimmunity.
The genetic condition DiGeorge Syndrome results in a small or absent thymus, which can lead to severe immunodeficiency and autoimmune diseases. Some medical treatments can also damage the thymus as a side effect. For example, chemotherapy and radiation therapy, which are common treatments for blood cancers like leukemia and lymphoma, not only deplete mature T cells but also significantly damage the thymus. Recovery of thymus function is required to generate new T cells after bone marrow transplantation and is delayed in some patients. This delay has been associated with increased risk of infections and poor clinical outcome.
- Current treatments for repairing the thymus
- How might stem cell research help?
- Lab-grown Thymus
- Find out more
- Acknowledgements and references
There are few clinically proven therapies available to improve or restore thymus function, but there are several directions of research:
- One current therapy is thymus transplantation, which can overcome the genetic absence of a thymus as in DiGeorge Syndrome. However, this treatment is only suitable for patients with no thymus or T cells, as the patients own T cells would attack and reject the transplant. Currently, only thymus fragments obtained from other, very young, patients as a by-product of heart surgery can be used for transplantation.
- A protein called ‘Interleukin-22’ can help thymuses that have been damaged by chemo or radio therapy to recover. There is a phase II clinical trial underway looking at the use of this protein as therapy for when bone marrow transplants are rejected by the body. The number of new T cells following treatment is being measured, which will provide evidence to whether the protein could be used as a treatment for boosting thymus function.
- Another protein called ‘Interleukin-7’ has shown very promising results in promoting immune regeneration in several pre-clinical models and in enhancing T cell counts in clinical studies. In humans, its direct effect on thymus recovery has yet to be fully investigated.
- Encouraging results from several clinical studies show that using human Growth Hormone enhances thymus recovery.
- Pre-clinical and clinical studies have shown that inhibiting a person’s production of sex hormones (sex steroids) either chemically or through surgery can promote thymus growth, resulting increased production of T cells.
Researchers have found stem or progenitor cells (stem cell-like cells) in the mouse thymus that can generate a thymus upon transplantation. Research is also looking at strategies to encourage better regeneration of the native thymus (in the body) and for engineering thymus tissue for transplantation. There are different sources of stem cells and approaches being used to do this:
1. Using tissue stem cells
If thymus cells from one mouse are transplanted into another mouse, they create a functional thymus that can produce T cells. Further work needs to be done to see if the same is true for human thymus progenitor cells. Also, before this approach can be clinically useful, scientists need to work out how to grow these thymus stem cells in the lab to make the numbers needed for transplantation.
2. Using pluripotent stem cells
Researchers have succeeded in making thymic-like cells from pluripotent stem cells in the lab. This work has been done with both mouse and human pluripotent cells. If these results can be improved on so that the lab-grown cells are identical to thymus cells in the body, it will pave the way for producing human thymus tissue in the lab for transplant. Making induced pluripotent stem cells from a patient’s own cells, and using these to create thymic cells, could present a way of overcoming the challenge of immune rejection.
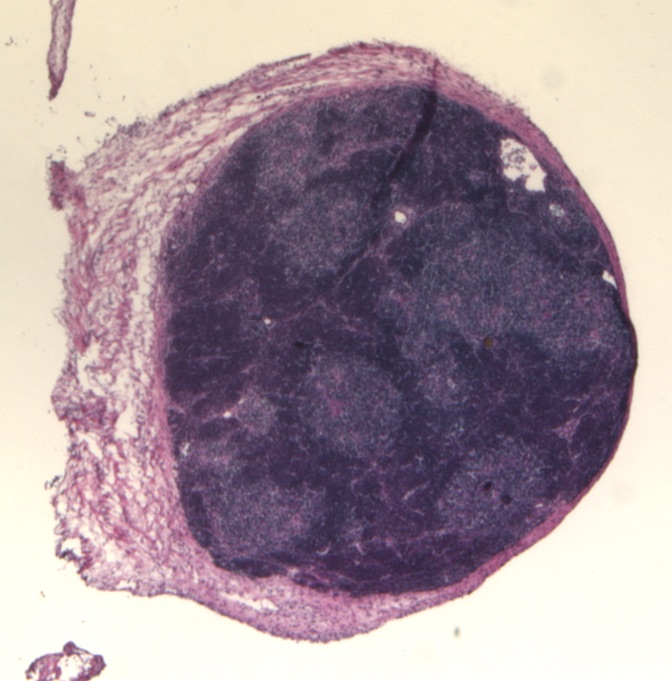
3. Reprogramming other tissue cells
Researchers have been able to make human ‘pre-T cells’ (the cells that will make and become T cells) from human blood progenitor cells in the lab, and transplantation of these cells in mice has shown promising results for speeding up the recovery of the immune system after bone marrow transplantation. The pre-T cells are transplanted at the time of therapy, and mature into T cells in the patient’s body.
Ongoing research has found a way to convert connective tissue cells, called fibroblasts, into thymus progenitor cells in the lab by a process called reprogramming. When transplanted into mice, these cells make a functioning thymus that supports normal T cell production. If this approach also works with human cells, these lab-grown cells may provide an alternative source of cells for thymus transplantation. Scientists are also using these lab-produced thymus cells to study thymus function, and to test improved ways of making a thymus or mini thymus (called an organoid) in the lab, that could be used for transplantation, and for testing new medicines.
Hopefully with additional time and resources, researchers will be able to develop ways to restore thymus function to help a range of patients.
Scientists have for the first time grown a complex, fully functional organ from scratch in a living animal by transplanting cells that were originally created in a laboratory. The team created a thymus - an organ in the body located next to the heart that produces immune cells known as T cells that are vital for guarding against disease. The advance could in future aid the development of 'lab-grown' replacement organs.
- Your amazing immune system interactive book http://www.interactive-immunity.net/
- 3D map showing the location of the thymus in humans http://www.healthline.com/human-body-maps/thymus
- Thymistem – EU funded research partnership using stem cell research to develop therapys for boosting the immune system http://www.thymistem.org/our-work
- Eurostemcell fact sheet on reprogramming cells http://www.eurostemcell.org/factsheet/cell-identity-and-reprogramming
- Eurostemcell fact sheet on induced pluripotent stem cells http://www.eurostemcell.org/factsheet/ips-cells-and-reprogramming-turn-any-cell-body-stem-cell
This factsheet was created by Amanda Holland, Emma Kemp, Cathy Southworth and Amy Hansen and reviewed by Clare Blackburn and Marcel R.M. van den Brink.
Images and videos
(copyright resides with the named contributor unless otherwise noted)
Illustration showing the location of the thymus in the human body © Cameron Duguid (CC BY 3.0)
Thymus organoid stained with Haemotoxylin and Eosin dyes © Nick Bredenkamp
Inside the thymus animation describing the function of the thymus © Cameron Duguid, Clare Blackburn, Emma Kemp, Cathy Southworth and Paul Rouse. (CC BY 3.0)
Lab grown thymus video describing how Clare Blackburn's laboratory grew a thymus organ in the lab © Medical Research Council